
Injecting Quality into Utilization Management
Estimated read time: 5 minutes
Here’s Something Many Organizations Didn’t See Coming
This isn’t a minor tweak. Starting with the 2026 NCQA Health Plan Accreditation standards, UM 1 now includes Elements B through H—a structured, closed-loop cycle of data reporting, analysis, committee oversight, and improvement action. That is the quality loop. It’s not enough to have a process. NCQA now wants to see whether your process is actually working.
For organizations pursuing First or Renewal surveys, this is one of the most operationally significant additions to the UM standards in years. If you haven’t started preparing, now is the time.
We’ve Heard the Concern
When we share these types of changes with clients, the reaction is often the same: a long pause, followed by “Wait—we have to report what?” There is so much angst and concern among our clients.
That reaction is understandable. Prior versions of the UM standards focused heavily on policies, procedures, criteria, and timeliness of decisions. What you measured internally was your business. The 2026 standards change that equation entirely.
As the saying goes—you can’t manage what you can’t measure. NCQA is now looking under the hood to understand what’s driving member dissatisfaction and whether organizations are taking meaningful action in response. The good news is that the requirements are structured and clear. The challenge is that they require coordination across UM operations, analytics, quality improvement, and governance—and they require data you may not yet be capturing in a reportable format. And it’s also a function that your UM committee may or may not be familiar with and may need some facilitation.
What Actually Changed in 2026
Let’s walk through the new framework so you know exactly what you’re working with.
The 2026 standards add a new data-driven oversight structure requiring organizations to:
- Report UM decision rates
- Analyze those rates internally
- Review the findings at the UM Committee level
- Implement improvement actions based on those findings
This creates a closed-loop evaluation process that connects UM operational data with governance and program improvement. Here’s a summary of the new elements:
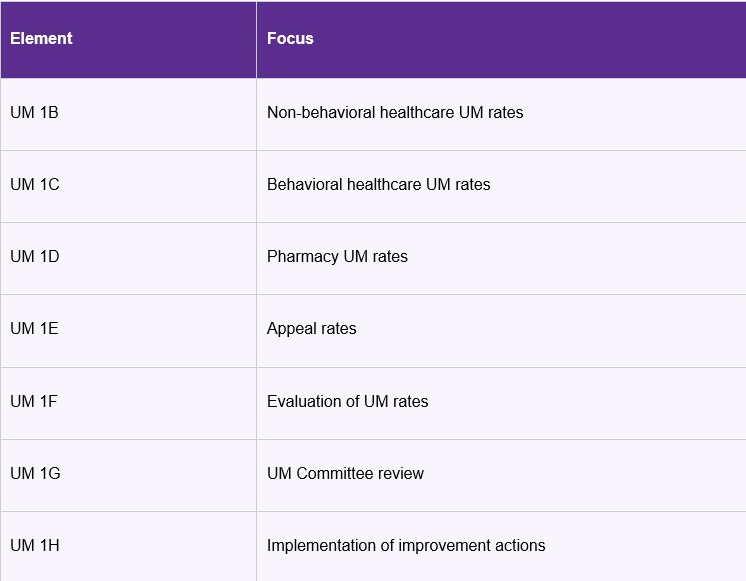
⚠ Important: Each of these elements is a structural requirement—defined by NCQA as an “essential program, process, and procedural component” that the organization is required to meet.
UM 1B–1D: New Reporting Requirements for Authorization Decisions
The most immediate operational change is the introduction of three new elements requiring annual reporting of UM authorization rates:
- UM 1B — Non-Behavioral Healthcare UM Rates
- UM 1C — Behavioral Healthcare UM Rates
- UM 1D — Pharmacy UM Rates
For each category, organizations must report seven metrics:
- Overall approval rate
- Overall denial rate
- Overall timeliness of notification rate for denials
- Timeliness of notification rate for urgent concurrent denials
- Timeliness of notification rate for urgent preservice denials
- Timeliness of notification rate for non-urgent preservice denials
- Timeliness of notification rate for post-service denials
The former UM 5 required metrics on timeliness, which were incorporated into the new UM 1, but this new requirement expands that into approval and denial rates.
A note on scoring: Factors 1 and 2—approval rate and denial rate—are critical factors. This means that if one critical factor is scored “No,” the element score cannot exceed Partially Met. If two or more critical factors are scored “No,” the element cannot exceed Not Met. Pay close attention to these!
Scoring Thresholds
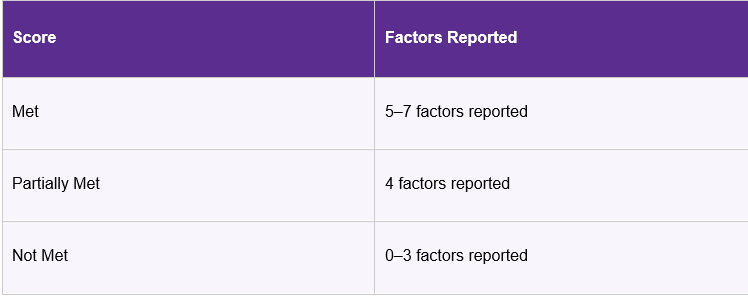
Organizations must report numerators and denominators for each metric using the NCQA UM Data Collection Workbook, based on 12 months of continuous data during the look-back period.
Important Clarifications About UM Rate Reporting
NCQA provides several clarifications that have real operational implications—and where we see organizations get tripped up.
Reporting Is Based on Requests, Not Services
Rates must be calculated at the authorization request level, regardless of how many services are included in a single request.
Partial Approvals and Denials
A request that is partially approved and partially denied counts in both the approval and denial calculations. Here’s a concrete example: a physical therapist requests 25 therapy visits. The plan approves 10 and denies 15. That single request counts as both a partial approval and a partial denial. Therefore, they need to be counted in both areas. Some systems will not be able to compute this accurately so clarify with your IT department whether that is doable.
Exclusions
Organizations must exclude the following from rate calculations:
- Withdrawn requests
- Pending decisions
- Duplicate requests
- Dismissed requests
This means your authorization tracking needs to accommodate these exclusions cleanly to produce a defensible rate for each required metric. Again, clarify with your IT department whether that is doable.
Timeliness Measurement
⚠ Common Misconception: Timeliness for denial notifications begins when the organization receives the authorization request—not when the relevant clinical information is received. Many plans believe the clock starts when the information comes in. It does not. If a NCQA selects this file and timeliness for that line of business has been exceeded from the receipt of the request to the decision date, you are at risk of missing a must-pass requirement.
UM 1E: New Appeal Rate Reporting
The 2026 standards also introduced UM 1E, which requires organizations to report two metrics:
- Overall appeal rate — how often denied requests are appealed
- Appeal overturn rate — how frequently those appeals result in a reversal of the original decision through internal or external review
Unlike authorization reporting, these appeal metrics do not require separate reporting by service category (behavioral health, pharmacy, etc.). However, pro tip: for internal decision making and transparency, you may want to segregate the categories. It also does not require the reasons for the appeal and for the overturn.
For optimal internal decision making, you may wish to report those also. For example, if there is a high rate for BH autism services appeals, and there is a high rate of overturn, you may want to consider withdrawal of the requirement for prior authorization, knowing that unnecessary hoops to jump through for the practitioner and member to delay services and engender dissatisfaction. This evaluation should be done (UM 1F) and sent to the UM Committee (UM 1G).
UM 1F: Required Evaluation of UM Rates
Reporting the data is only the first step. Under UM 1F, organizations must conduct an annual evaluation of all reported UM rates—non-behavioral, behavioral, pharmacy, and appeal.
The evaluation must assess whether the rates are appropriate relative to the organization’s own operations. That assessment may include comparisons to internal historical data or external benchmarks such as state or federal analyses. This evaluation does not require the full quantitative and qualitative analysis (although it would never hurt) but you are required to draw a conclusion.
Key Point: NCQA does not determine whether rates are appropriate. That determination must come from the organization’s own analysis. This is where your analytical team and clinical leadership need to be engaged.
UM 1G: UM Committee Oversight
The evaluation doesn’t stay with the analytics team—it goes to governance. Under UM 1G, the UM Committee must annually:
- Review the UM rate evaluation
- Evaluate the overall UM program
- Identify improvement opportunities and recommend actions
The committee’s review should consider whether the UM program remains appropriate, including its structure and scope, UM criteria, prior authorization requirements, UM decision patterns, and physician and behavioral health practitioner oversight.
Meeting minutes must clearly document the discussion and any recommended actions. This is not the place for vague minutes. Surveyors will be looking for evidence of a substantive review.
UM 1H: Implementation of Improvement Actions
Finally, UM 1H requires organizations to demonstrate that improvement actions were actually implemented—not just discussed. Organizations must show evidence of implementing at least one recommendation related to the UM program or UM rates.
Examples might include:
- Revising prior authorization requirements
- Adjusting UM workflows to improve timeliness
- Enhancing staff training related to denial notifications
- Modifying criteria application processes
A Word of Caution About “Backfilling”
For organizations undergoing a survey in 2026, the pressure is on to not only gather the data and report the metrics—but to evaluate the data and demonstrate that at least one metric drove a remediation action. When these types of requirements are first implemented, we often see “backfilling”: an organization is already undertaking some initiative and tries to link it retroactively to a metric and a standard. Surveyors are experienced and look for a logical connection between the reported metrics and the improvement action. The linkage needs to be clear and credible.
When These Requirements Apply
The new UM 1 elements apply to:
- First Surveys
- Renewal Surveys
They are not applicable to Interim surveys. Organizations must complete the UM Data Collection Workbook at least once during the year prior to the survey. The workbook is available from NCQA for organizations that have applied for accreditation.
Why This Shift Matters
These changes represent an important evolution in how NCQA evaluates UM programs. Historically, accreditation reviews focused heavily on process compliance—do you have the right policies? Are your decisions timely? The 2026 standards now require organizations to analyze the outcomes of those processes, particularly how authorization decisions and appeals function in practice.
For many organizations, this will require stronger coordination between UM operations, analytics teams, quality improvement staff, and governance committees. Health plans that begin preparing early will be better positioned to ensure the necessary data capture, reporting structures, and committee review processes are in place before their next accreditation survey.
How MHR Helps Health Plans Prepare
At MHR, we have been helping health plans operationalize the new UM 1 requirements for several months – as soon as the 2026 standards were published. Our team—several of whom are current or former NCQA surveyors—helps organizations translate the standards into practical, defensible operational processes.
- Need to build your UM data reporting process from the ground up? We’ve done it.
- Not sure how to structure your UM Committee documentation to reflect the new requirements? We can guide you.
- Want to make sure the link between your UM rate analysis and your improvement actions is clear and credible? That’s exactly the kind of gap we’re trained to find.
✅ Ready to prepare for the 2026 UM standards? We understand that it’s a big shift in how things are done for UM.
To help you get started, we created a UM Rate Reporting Readiness Checklist your team can use to quickly evaluate whether your organization is prepared for the 2026 UM 1 requirements.
Connect with MHR to ensure your UM program, reporting structure, and committee governance processes are aligned with NCQA expectations. Reach us at [email protected] or visit managedhealthcareresources.com.
MHR — Driving healthcare quality one NCQA accreditation at a time.



